How Kidney C.O.P.® Works
How Does Kidney C.O.P.® Protect Against Calcium Oxalate Crystals?
Kidney C.O.P.® is formulated specifically to help reduce calcium oxalate stones in the kidney.
Scientists have studied the ingredients in Kidney C.O.P.® and suggest they work by:
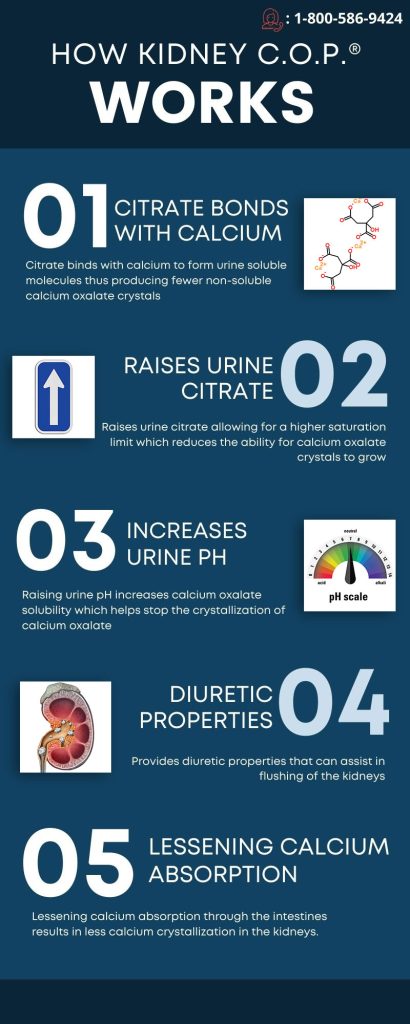
- – Calcium oxalate solubility – Citrate binding with calcium to form urine soluble molecules and producing fewer non soluble calcium oxalate crystals**
- – Raising urine citrate allowing for a higher saturation limit reducing the ability for calcium oxalate crystals to grow (which leads to crystallization in the kidneys)**
- – Raising urine pH increases calcium oxalate solubility which inhibits the crystallization of calcium oxalate in the kidney**
- – Providing diuretic properties that can assist in the flushing of the kidneys and the dissolution of calcium oxalate**
- – Lessening absorption of calcium through intestines resulting in less calcium crystallizing in the kidney**
- – Using delayed release vegetarian capsule to avoid dissolving in the stomach**
Learn more about calcium oxalate in the kidneys and how Kidney C.O.P.® can help:
Calcium oxalate crystals can form in the kidney over time and can cause significant discomfort. While this condition can happen to many people, this is often a recurring problem for some people. Oxalates are compounds found in a wide variety of foods. When the oxalates bind to the calcium in your kidneys, the kidney calcium oxalate crystals start to build up and have the potential to cause problems.
Kidney C.O.P.® is a natural supplement formulated by a medical doctor, pharmacist, and pharmaceutical professionals. Its truly unique formula utilizes FDA GRAS (generally recognized as safe) ingredients and is GMO-free, preservative free, sugar free, and gluten free. The Kidney C.O.P.® formulation also does not contain potential problematic ingredients like dairy, soy, nuts, or fish. Most importantly there’s no need for a prescription.
Finally, Kidney COP® utilizes a patented, delayed release capsule that overcomes the problem of the capsules totally dissolving in your stomach. It is thought to provide better absorption and help get more of the active ingredients working for your kidneys. Without a delayed release capsule, the ingredients that facilitate all the benefits of Kidney C.O.P.® would not be maximized.